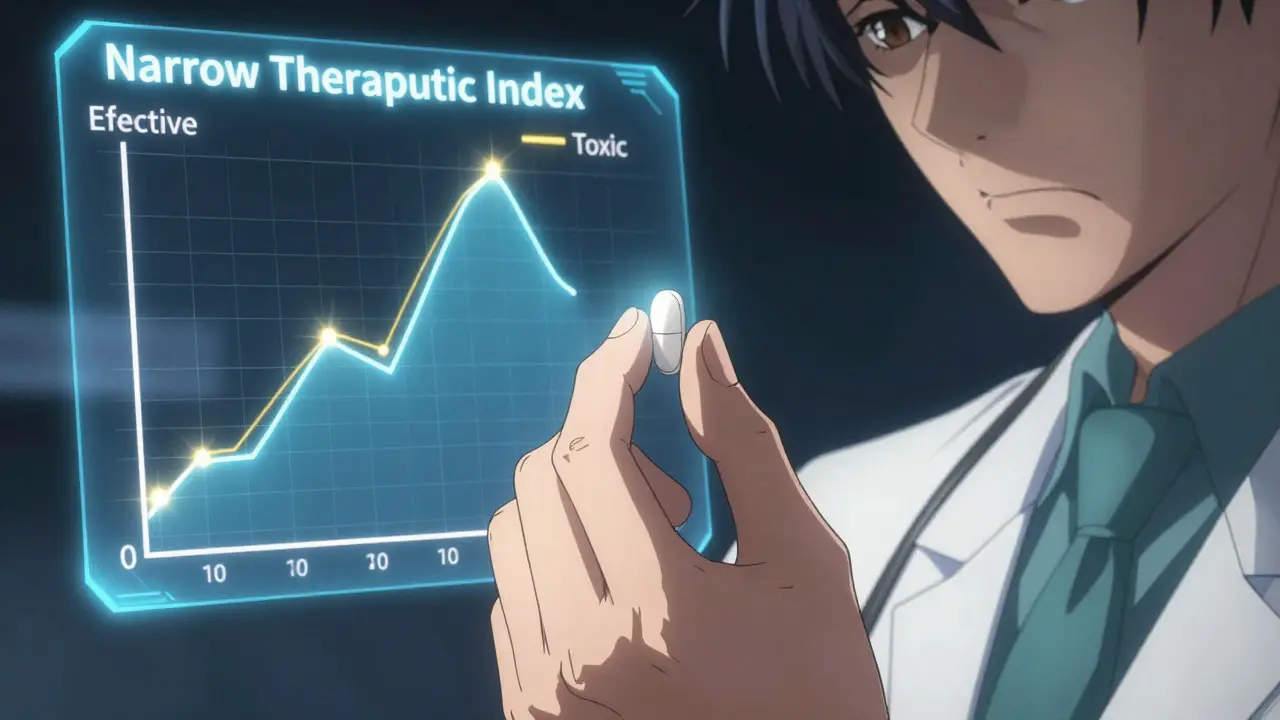
Imagine prescribing a medication where a tiny shift in dosage-something as small as a few micrograms-could be the difference between a patient staying healthy or ending up in the emergency room. This is the high-stakes reality of Narrow Therapeutic Index is a category of drugs where the margin between an effective dose and a toxic dose is extremely slim. Commonly referred to as NTI drugs, these medications require precision that most standard drugs simply don't. Because the risk is so high, a long-standing tension exists between doctors who want total control over the brand and pharmacists who aim to lower costs through generic substitution.
The Bioequivalence Battle: Why Prescribers Hesitate
The core of the conflict lies in bioequivalence. For most drugs, the FDA allows a relatively wide range for generic versions to be considered "the same" as the brand name. However, for NTI drugs, that gap is too dangerous. To address this, the FDA is the federal agency responsible for protecting public health by ensuring the safety, efficacy, and security of human and veterinary drugs which implemented stricter guidelines in 2019. They narrowed the acceptable bioequivalence range to 90-111%, compared to the usual 80-125%.
Despite these tighter rules, many doctors aren't convinced. Specialized practitioners, particularly those in transplant medicine, often argue that testing generics on healthy volunteers isn't enough. They believe these drugs should be tested in actual patients to see how they behave in a diseased system. This skepticism leads many prescribers to use "dispense as written" (DAW) instructions to prevent any switching, especially when a patient has finally found a stable dose.
How Different Specialists View Substitution
Not all doctors feel the same way about switching to generics. The level of concern usually scales with the volatility of the drug being prescribed. For instance, Warfarin is an anticoagulant medication used to prevent blood clots, known for its narrow therapeutic window and requirement for frequent INR monitoring is a major point of contention. About 68% of physicians have expressed anxiety over generic substitutions for this drug, fearing that fluctuations in the International Normalized Ratio (INR) could lead to dangerous bleeding or clotting events.
Psychiatrists managing patients on Lithium is a mood stabilizer used primarily to treat bipolar disorder, requiring strict serum level monitoring to avoid toxicity also show higher caution. In fact, psychiatrists receive nearly double the amount of substitution notifications compared to primary care physicians, reflecting the sensitivity of mood-stabilizing regimens.
| Drug Name | Primary Use | Brand Persistence % |
|---|---|---|
| Tacrolimus | Transplant immunosuppression | 32% |
| Warfarin | Blood thinning (Anticoagulant) | 28% |
| Levothyroxine | Thyroid hormone replacement | 25% |
| Phenytoin | Seizure control (Anticonvulsant) | 21% |
| Lithium | Bipolar disorder stabilization | 19% |

The Regulatory Patchwork Across the U.S.
Depending on where a patient lives, the rules for substitution change. Currently, about 28 U.S. states have specific laws regarding NTI drugs. In places like Texas and Florida, official lists restrict automatic substitution, meaning the pharmacist must either notify the prescriber or get their consent first. This legal safety net gives doctors a sense of control that they lack in "open" states.
Interestingly, states that require "affirmative patient consent" see significantly lower generic substitution rates-roughly 23% lower than states without these laws. This suggests that when patients are told about the switch, they often prefer to stick with what they know, likely influenced by their doctor's cautious tone. Meanwhile, organizations like the AMCP is the Academy of Managed Care Pharmacy, an organization focused on optimizing patient outcomes through the responsible use of medications generally oppose these restrictions, arguing that scientific evidence supports the use of generics when managed by a professional pharmacist.

The Communication Gap: Phone Calls vs. Portals
One of the biggest headaches for prescribers isn't necessarily the drug itself, but the workflow. When a pharmacist substitutes an NTI drug, the doctor needs to know. But how? Most doctors hate the traditional phone call. Research shows that 63% of physicians prefer electronic notifications. Why? Because a phone call interrupts a patient visit, whereas an electronic alert can be reviewed during a designated chart-review period.
When communication fails, the consequences are real. The ISMP is the Institute for Safe Medication Practices, a nonprofit organization dedicated to preventing medication errors has tracked thousands of NTI-related errors. While only a small fraction result in serious harm, the administrative burden is heavy. Roughly 29% of doctors report an increase in office visits just to monitor patients after a generic switch, adding unnecessary costs and stress to both the provider and the patient.
Economic Trade-offs and Future Outlook
There is a massive financial tug-of-war happening in the background. On one side, the Congressional Budget Office suggests that restricting generic substitution could cost Medicare over $1 billion annually. On the other side, proponents of generics argue that wider adoption could save the healthcare system over $120 billion over a decade.
Are doctors becoming more open to generics? The data suggests yes, but slowly. New evidence from the ASCO is the American Society of Clinical Oncology, the leading professional organization for oncology care now supports generic substitution for certain oral oncology drugs, provided there is strict therapeutic drug monitoring. As real-world evidence from trials like PRESCRIPT-NTI continues to roll out, the "fear factor" surrounding generics may decrease.
By 2028, analysts predict generic penetration for NTI drugs will hit 78%. This shift won't happen because the drugs suddenly become "safer," but because the systems for communicating substitutions are becoming more efficient. When a doctor trusts the notification system, they are more likely to trust the generic pill.
What exactly makes a drug "Narrow Therapeutic Index"?
A drug is classified as NTI when the difference between the dose that works and the dose that is toxic is very small. Technically, the FDA looks for a ratio between the minimum toxic concentration and the minimum effective concentration that is less than or equal to two.
Why do some doctors insist on brand-name NTI drugs?
Mainly due to stability concerns. Even though generics are bioequivalent, small variations in absorption can lead to a patient's blood levels fluctuating, which can cause a relapse in condition or toxic side effects.
Do generics of NTI drugs have different FDA standards?
Yes. Since 2019, the FDA has required a tighter bioequivalence range (90-111%) for NTI generics compared to the standard 80-125% range used for most other generic medications.
How can pharmacists and doctors better coordinate substitutions?
The shift toward electronic notifications is the most effective method. Moving away from phone calls to standardized digital alerts allows doctors to track changes in a patient's medication history more accurately.
Which NTI drugs are most commonly kept as brand names?
Tacrolimus, Warfarin, and Levothyroxine typically show the highest brand-name persistence, as these medications are critical for organ transplant success, blood clotting control, and thyroid function.